Arbormed's "unique" Wilson disease mechanism, safety and eff…
페이지 정보

본문
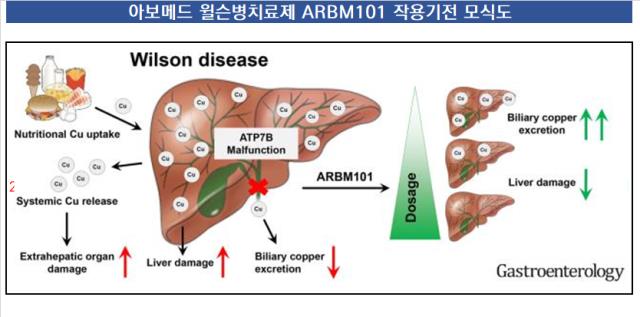
The unique mechanism of "ARBM-101," which Arbormed is developing as a treatment for Wilson's disease, an intractable rare disease, has been demonstrated through preclinical examination. Wilson's disease is a disease in which copper accumulated in the body is not discharged and causes various liver diseases and complications. ARBM-101 has demonstrated the effectiveness of the new mechanism by taking this pathway to the rectum, i.e., feces, in discharging accumulated copper.
The previously released Wilson disease treatment was developed to prevent additional copper from accumulating in the liver. However, in this process, various side effects such as kidney risk (renal failure) are concerned while using the discharge route as urinary tract, that is, urine. If Avomed proves the natural emission mechanism of copper with ARBM-101, it will present a new possibility for overcoming Wilson's disease, which has no fundamental treatment 110 years after its discovery.
◇ Confirmation of preclinical pharmacological efficacy of 'copper stool discharge' using microorganisms of 'ARBM-101'
Arbormed recently published a paper containing the results of animal experiments in the official journal of the American Society of Digestive Sciences, "Gastroenterology." The title of the paper is 'ARBM101 discharges excessive liver copper through bile excretion in mice that caused Wilson's disease. The main point is to prove the efficacy of ARBM-101 as a disease animal model based on copper emissions, an important evaluation variable for the treatment of Wilson's disease.
Arbormed is developing ARBM-101 as a microbial-based treatment for rare diseases. The microorganism was introduced by the Helmholtz Research Institute in Germany, which is a Noble target in the form of microorganisms selectively binding to copper in the body and then discharging it through feces, and the first therapeutic mechanism in the family.
Wilson's disease, a rare disease under development by Arbormed, is a disease in which copper accumulated in the body is not discharged due to genetic abnormalities. It is mainly accumulated and infiltrated into the liver, causing complications of various liver diseases. Among rare diseases, it is considered one of the pillars with a relatively high incidence rate, but there is no fundamental treatment even though it has been 110 years since the disease was known.
All it takes is a successful initial diagnosis and a liver transplant operation. Penicillin, which prevents further copper accumulation through diuretics, was approved by the U.S. Food and Drug Administration in 1970, but 30% of patients stop drug treatment due to relatively low symptom relief and various side effects including kidney.
ARBM-101 is a mechanism that efficiently exports copper through feces, focusing on the selective adhesion of microorganisms to copper. To date, we have demonstrated that administering ARBM-101 to mice that caused Wilson's disease induces rapid copper excretion compared to the control group. The pharmacological effect of minimizing side effects by discharging copper through feces, a physiological phenomenon, was confirmed.
◇ A total of 12 advisory committee members are experts in Wilson's disease... The first step into the global top prize next year
Arbormed plans to develop ARBM-101 as an innovative new drug that will change the paradigm of Wilson's disease treatment. Based on the results of the animal experiment, it is expected that it will be able to enter the first phase of global clinical trials as early as next year and secure research data.
As the mechanism is excellent and the risk of side effects is low, U.S. Wilson's disease specialists are also showing a lot of interest in Arbormed's ARBM-101. The company recently formed a total of 12 clinical advisory committees centered on these specialists and began to work together to launch the first new drug in the group.
Yale University professor Michael Schilsky, who created guidelines for treating Wilson's disease in detail, is drawing attention as he chairs the advisory committee. In addition, five of the top seven local hospitals designated by the Wilson Disease Association (WDA) as capable of treating Wilson's disease will participate.
Arbormed previously released a study by the American Liver Society (AASLD) in 2022 that the administration of ARBM-101 to animal models has the effect of promoting rapid discharge of copper while minimizing side effects. At that time, one of the Abomed advisors, Professor Valentina Medici Yoo Davis, was in charge of the presentation.
Following the presentation of ARBM-101 animal model research data at major conferences last year, we have recently established a clinical advisory committee network for renowned medical staff in the United States, said Park Kyo-jin, CEO of Arbormed.
Meanwhile, Arbormed was founded in 2017 by Park Kyo-jin, co-founder of MedCI, a new drug licensing consulting company. Lim Won-bin, a former head of the Dong-A Pharmaceutical Research Institute, joined the group in 2020 to serve as co-chairman. In addition to Wilson's disease, it is developing treatments for rare diseases such as acute respiratory distress syndrome. Since its establishment, about 27 billion won has been raised from FIs such as Ildong Pharmaceutical and VC.
The previously released Wilson disease treatment was developed to prevent additional copper from accumulating in the liver. However, in this process, various side effects such as kidney risk (renal failure) are concerned while using the discharge route as urinary tract, that is, urine. If Avomed proves the natural emission mechanism of copper with ARBM-101, it will present a new possibility for overcoming Wilson's disease, which has no fundamental treatment 110 years after its discovery.
◇ Confirmation of preclinical pharmacological efficacy of 'copper stool discharge' using microorganisms of 'ARBM-101'
Arbormed recently published a paper containing the results of animal experiments in the official journal of the American Society of Digestive Sciences, "Gastroenterology." The title of the paper is 'ARBM101 discharges excessive liver copper through bile excretion in mice that caused Wilson's disease. The main point is to prove the efficacy of ARBM-101 as a disease animal model based on copper emissions, an important evaluation variable for the treatment of Wilson's disease.
Arbormed is developing ARBM-101 as a microbial-based treatment for rare diseases. The microorganism was introduced by the Helmholtz Research Institute in Germany, which is a Noble target in the form of microorganisms selectively binding to copper in the body and then discharging it through feces, and the first therapeutic mechanism in the family.
Wilson's disease, a rare disease under development by Arbormed, is a disease in which copper accumulated in the body is not discharged due to genetic abnormalities. It is mainly accumulated and infiltrated into the liver, causing complications of various liver diseases. Among rare diseases, it is considered one of the pillars with a relatively high incidence rate, but there is no fundamental treatment even though it has been 110 years since the disease was known.
All it takes is a successful initial diagnosis and a liver transplant operation. Penicillin, which prevents further copper accumulation through diuretics, was approved by the U.S. Food and Drug Administration in 1970, but 30% of patients stop drug treatment due to relatively low symptom relief and various side effects including kidney.
ARBM-101 is a mechanism that efficiently exports copper through feces, focusing on the selective adhesion of microorganisms to copper. To date, we have demonstrated that administering ARBM-101 to mice that caused Wilson's disease induces rapid copper excretion compared to the control group. The pharmacological effect of minimizing side effects by discharging copper through feces, a physiological phenomenon, was confirmed.
◇ A total of 12 advisory committee members are experts in Wilson's disease... The first step into the global top prize next year
Arbormed plans to develop ARBM-101 as an innovative new drug that will change the paradigm of Wilson's disease treatment. Based on the results of the animal experiment, it is expected that it will be able to enter the first phase of global clinical trials as early as next year and secure research data.
As the mechanism is excellent and the risk of side effects is low, U.S. Wilson's disease specialists are also showing a lot of interest in Arbormed's ARBM-101. The company recently formed a total of 12 clinical advisory committees centered on these specialists and began to work together to launch the first new drug in the group.
Yale University professor Michael Schilsky, who created guidelines for treating Wilson's disease in detail, is drawing attention as he chairs the advisory committee. In addition, five of the top seven local hospitals designated by the Wilson Disease Association (WDA) as capable of treating Wilson's disease will participate.
Arbormed previously released a study by the American Liver Society (AASLD) in 2022 that the administration of ARBM-101 to animal models has the effect of promoting rapid discharge of copper while minimizing side effects. At that time, one of the Abomed advisors, Professor Valentina Medici Yoo Davis, was in charge of the presentation.
Following the presentation of ARBM-101 animal model research data at major conferences last year, we have recently established a clinical advisory committee network for renowned medical staff in the United States, said Park Kyo-jin, CEO of Arbormed.
Meanwhile, Arbormed was founded in 2017 by Park Kyo-jin, co-founder of MedCI, a new drug licensing consulting company. Lim Won-bin, a former head of the Dong-A Pharmaceutical Research Institute, joined the group in 2020 to serve as co-chairman. In addition to Wilson's disease, it is developing treatments for rare diseases such as acute respiratory distress syndrome. Since its establishment, about 27 billion won has been raised from FIs such as Ildong Pharmaceutical and VC.
관련링크
- 이전글[Complex Generic] Arbormed signs Copyright Agreement with US and Indian Pharmaceutical Companies 23.06.07
- 다음글[Clinical Advisory Committee] Arbormed's Wilson's disease treatment attracts global attention with the emergence of Clinical Research Avengers 23.05.03
댓글목록
등록된 댓글이 없습니다.