Diversified pharmaceutical company with divisions
for novel medicines and complex generics
SCROLL >
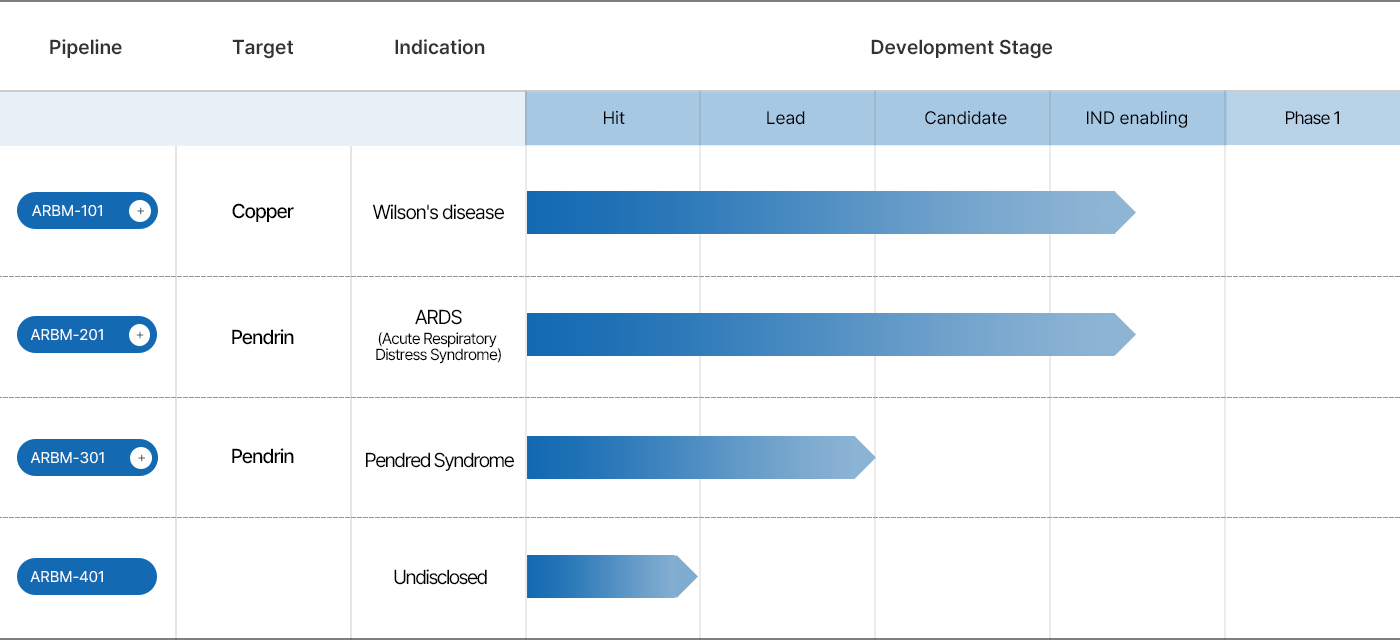

| TARGET | INDICATION | DEVELOPMENT STAGE |
|---|---|---|
| Copper | Wilson’s disease | IND-enabling studies Preclinical stage: - GLP toxicology study ongoing - IND submission: Q2 2026 - Phase 1a initiation: Q3 2026 |
Copper
Wilson’s disease
IND-enabling studies
Preclinical stage:
- GLP toxicology study ongoing
- IND submission: Q2 2026
- Phase 1a initiation: Q3 2026
ARBM-101 is expected to prevent or stop WD liver damage from worsening by facilitating copper excretion as it was confirmed in a WD rat model. Its unprecedented de-coppering efficacy in WD animals presents its superior therapeutic potential that none of the currently available copper chelators can achieve.

The image modified from https://www.osmosis.org/learn/Wilson_disease
ARBM-101 prevented or stopped the progression of copper-overloaded liver damage by eliminating excess copper from the liver into feces in WD rats. Such an efficacy is thought to bedriven by its enormously high affinity to copper and by its facilitation of excretion through bile.

Modified from Ala A. et al. 2007; The Lancet. & Patil M. et al. 2013; J. Clin. Exp. Hepatol. & Polishchuk E.V. et al. 2019; Gastroenterol.
A methanobactin peptide was shown to be superior to any other copper-chelating drugs or drug candidates in terms of the ability of reducing liver copper levels by facilitating excess copper excretion as shown in the remnant copper contents post treatment upon i.p. injections to WD rats (left panel). Methanobactins facilitate copper excretion through bile-fecal path as directly demonstrated by liver perfusion experiments (right panel). Such mode of action makes them markedly distinct from the currently marketed drugs such as D-PA or Trientine (TETA), both of which chelate blood copper, rendering chelated copper ions passively excreted into urine. It was also evident that methanobactins were superior to tetrathiomolybdate (TTM) in the excretion levels among those mainly taking bile-fecal path (right panel).

Lichtmannegger, J., et al., 2016; J. Clin. Invest.
Hepatocytic mitochondria were shown to recover their activities including their capability of producing ATP upon ARBM-101 treatment in a WD rat model (Data not shown). Although the mechanism of ARBM-101 rescuing mitochondrial function is still under investigation, it is certain that prompt elimination of excess copper from the cell by ARBM-101 allows mitochondria to recover their functionality leading to prevention of hepatocytic cell death and of liver disease progression.


| TARGET | INDICATION | DEVELOPMENT STAGE |
|---|---|---|
| Pendrin, PDS | ARDS (Acute Respiratory Distress Syndrome) |
IND-enabling studies Preclinical stage: - IND submission: Q4 2026 - Phase 1 initiation: Q1 2027 |
Pendrin, PDS
ARDS
(Acute Respiratory Distress Syndrome)
IND-enabling studies
Preclinical stage:
- IND submission: Q4 2026
- Phase 1 initiation: Q1 2027
ARBM-201 has shown a therapeutic potential to treat acute lung injury (ALI) by reducing the expression of pendrin and its anion exchange activity in the alveolar epithelial cells and blocking the downstream signaling pathway and inflammation, as confirmed in an LPS-induced ALI mice model. Since the pendrin expression is increased in both LPS-treated mouse airways and patients with ARDS caused by pneumonia, it strongly suggests a high possibility for the clinical application of pendrin inhibitor (ARBM-201) in inflammatory airway disease.

ARBM-201 is a new chemical entity with high selectivity for the SLC26A4 gene, which encodes a protein called pendrin. As a pendrin inhibitor, ARBM-201 binds to pendrin, blocks the transepithelial transport of SCN- and then inhibits OSCN- generation and NF-κB activation, which results in the suppression of proinflammatory cytokine production, neutrophil infiltration and subsequent lung injury.

Modified from Lee, E. H., et al., 2020; Theranostics.
Pendrin expression level increased in LPS-treated mouse airways and also in BALF from pneumonia-induced ARDS patients (Top). While the total cell count and protein concentration in bronchoalveolar lavage fluid was markedly increased after LPS treatment in the wild-type mice, they did not in pendrin-null mouse (Bottom left). In addition, simultaneous application of NaSCN with LPS-induced robust lung injury in pendrin-null mice, whereas administration of LPS alone did not induce Acute Lung Injury(ALI) (Bottom right). These data strongly indicate the critical role of pendrin in ALI pathogenesis and that airway surface SCN- transported by pendrin is an essential component for LPS-induced airway inflammation.

Lee, E. H., et al., 2020; Theranostics.
Treatment of pendrin inhibitor after LPS administration significantly reduced BALF total cell count and protein concentration, lung injury score, phospho-IκB protein expression, as well as proinflammatory cytokine levels in the LPS-induced ALI mice model.

Lee, E. H., et al., 2020; Theranostics.

| TARGET | INDICATION | DEVELOPMENT STAGE |
|---|---|---|
| Pendrin | Pendred Syndrome |
Candidate selection stage - Development of a Pendrin corrector - Preclinical development ongoing |
Pendrin
Pendred Syndrome
Candidate selection stage
- Development of a Pendrin corrector
- Preclinical development ongoing
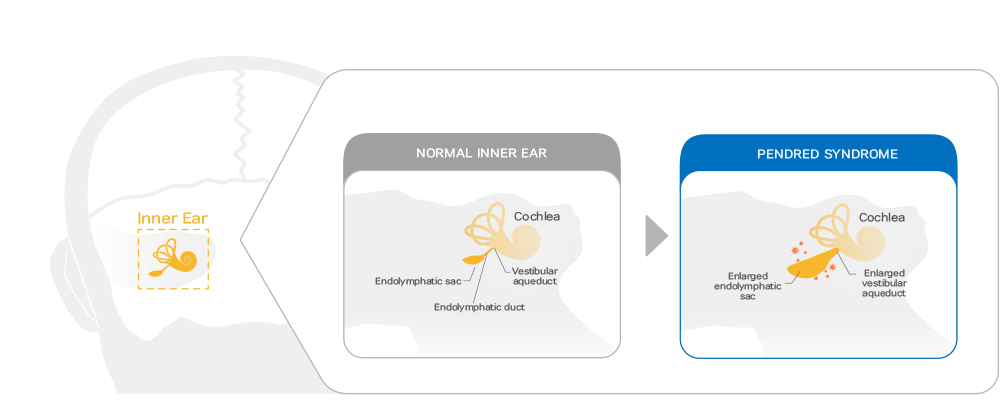
Pendred syndrome is inherited in an autosomal recessive manner by genetic mutations on SLC26A4. The structural abnormality caused by genetic mutations on SLC26A4 prevents pendrin protein expression on the cell surface, which results in pH disturbance within the inner ear, leading to hearing loss. The structure of pendrin protein corrected by ARBM-301 makes its surface expression restored and its function also rescued as an anion exchanger.
ARBM-301 is designed to rescue pendrin protein from degradation by correcting its conformation of the mutated pendrin protein retained in the ER thereby allowing its targeting to the cell surface with further post-translational modifications in the TGN on the way.
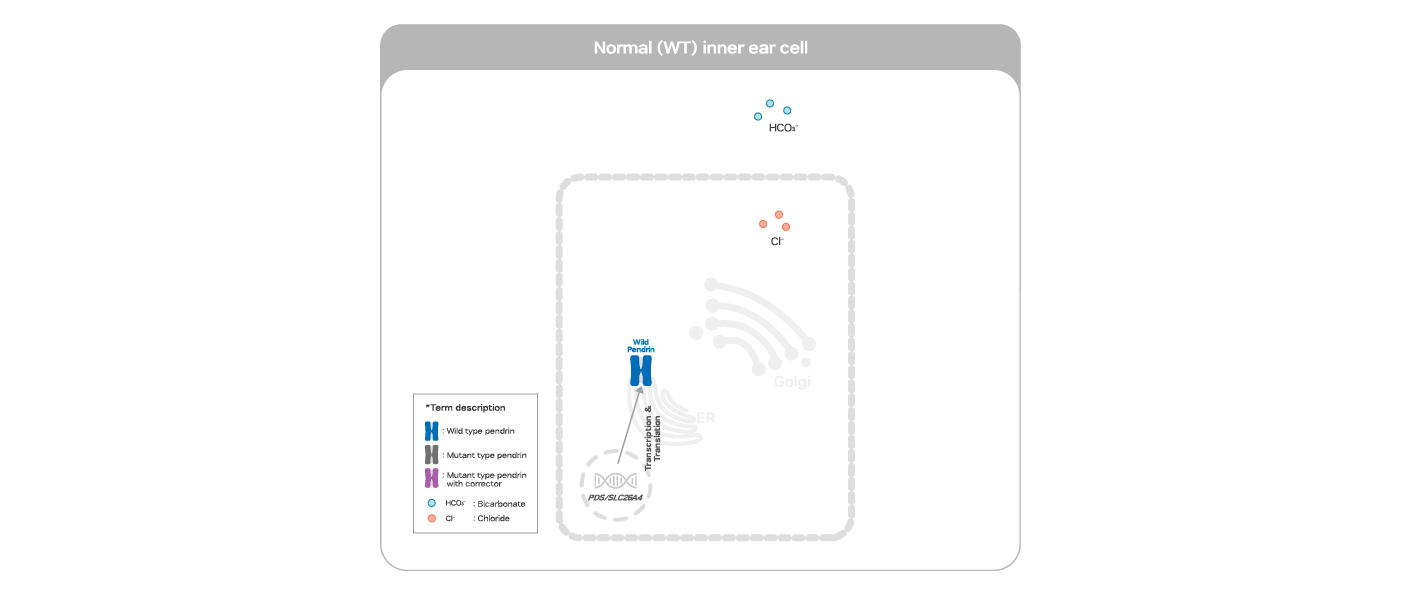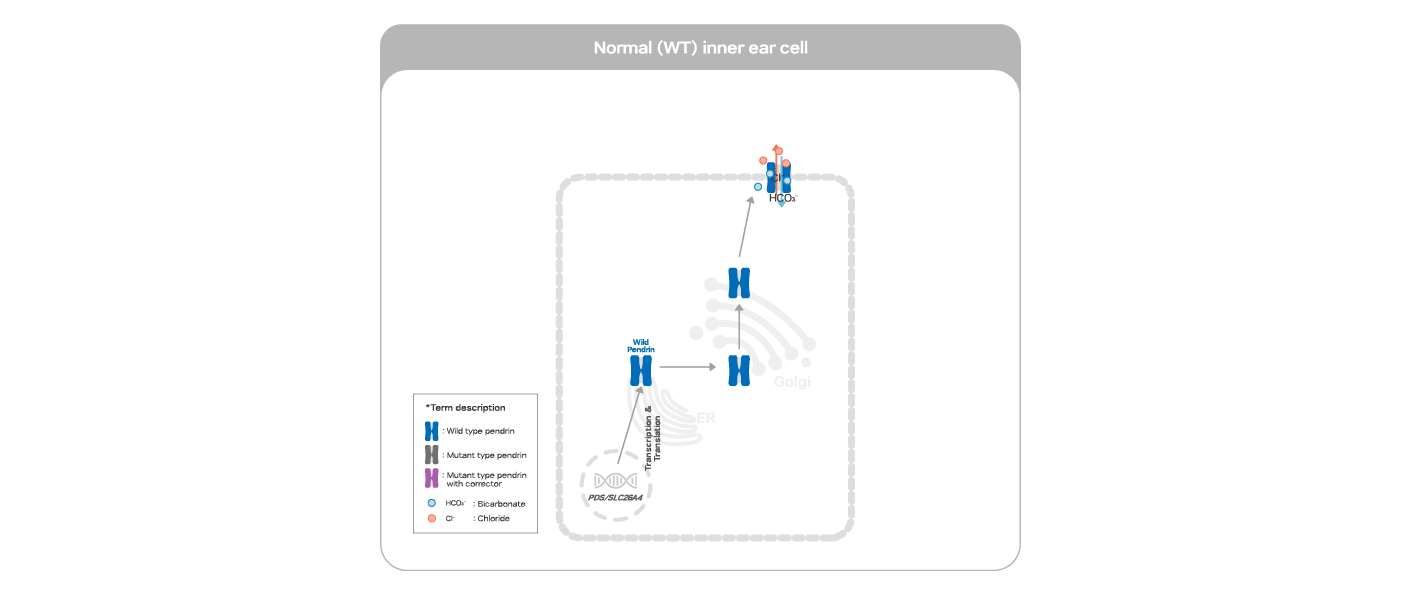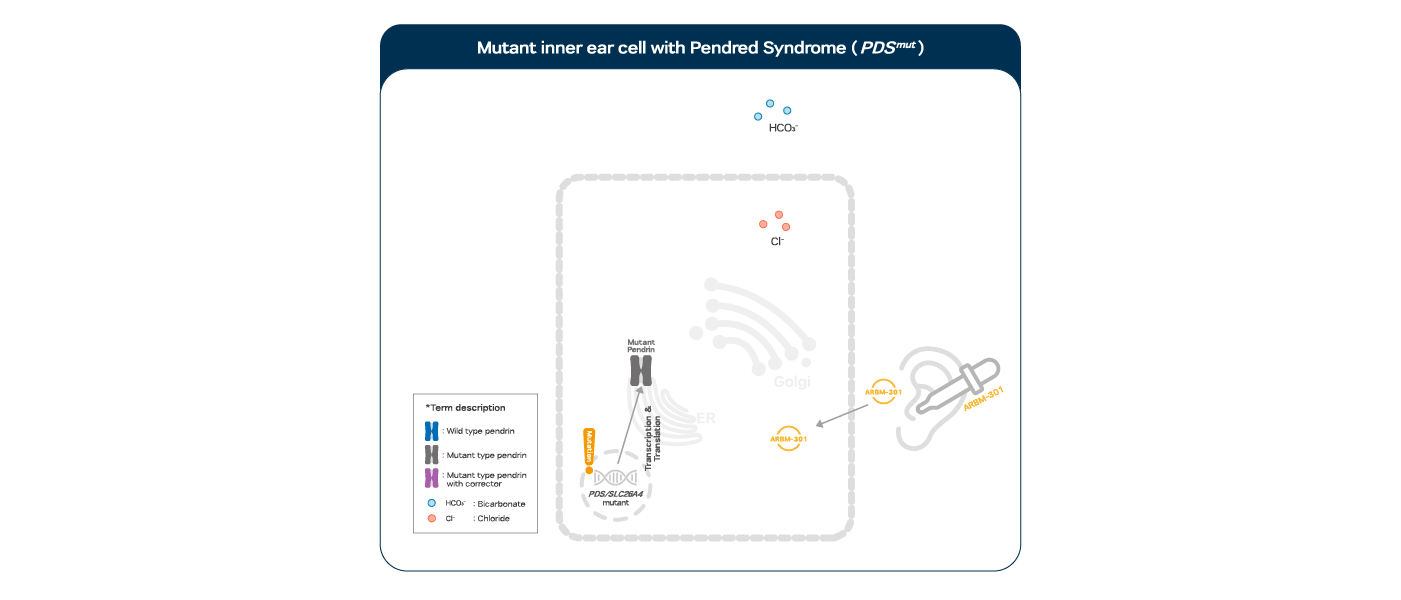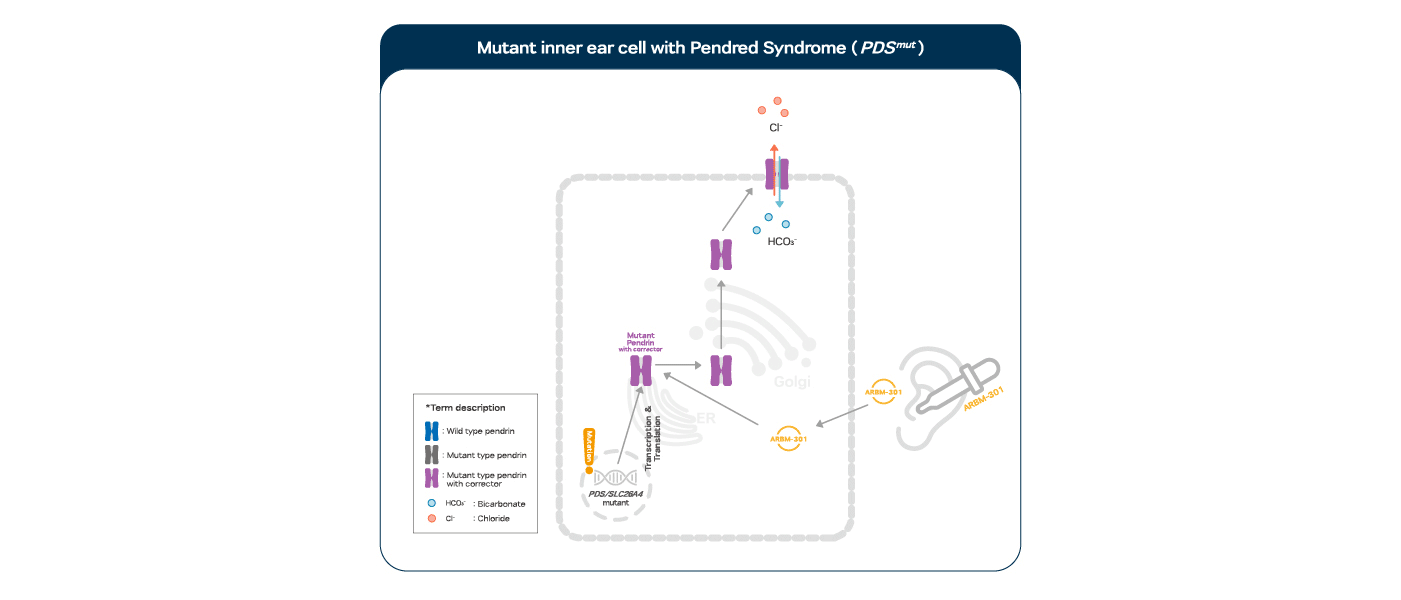
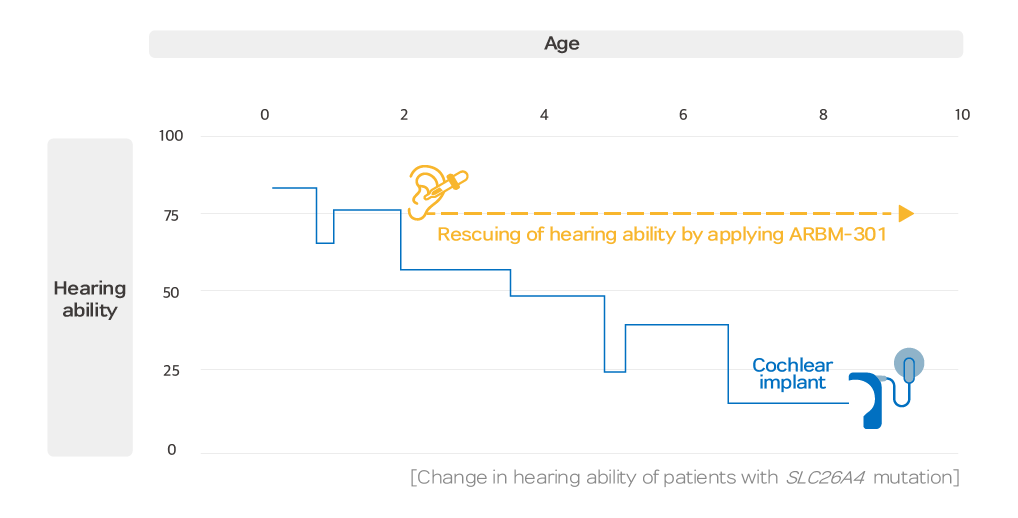
The rescuing effect of pendrin correctors on the disabled hearing of the patients with SLC26A4 mutations.