[Clinical Advisory Committee] Arbormed's Wilson's disease treatment at…
페이지 정보

본문
Arbormed, a domestic biopharmaceutical company, has formed a clinical advisory committee comprising 12 American Wilson's disease specialists to support the clinical trials of our pipeline, ARBM-101. This is a significant move for a domestic biopharmaceutical company, indicating the high anticipation of ARBM-101 in overseas medical fields. Professor Michael Schilsky of Yale University, who created the guidelines for the treatment of Wilson's disease, will serve as the advisory chairman, while five of the top seven local hospitals designated by the Wilson's Disease Association (WDA) as capable of treating Wilson's disease will participate.
It is difficult to find a case in which this level of advisory committee has been formed to support clinical trials of new drug pipelines by domestic bio ventures. This is proof that overseas medical sites have high expectations for ARBM-101.
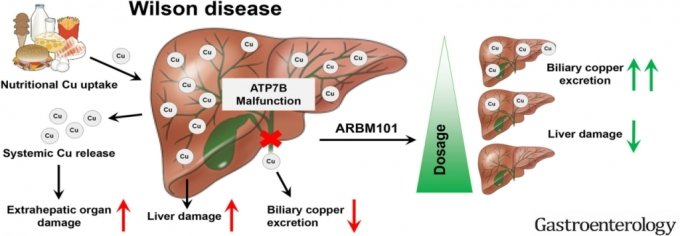
Arbormed recently published a study in an international journal demonstrating the safety and effectiveness of ARBM-101.
In March, the results of an animal model experiment of ARBM-101 were published in Gastroenterology (IF 33.883), a world-renowned journal in the field of digestive organs and the official journal of the American Society of Gastroenterology.
The title of this paper is <ARBM101 (Methanobactin SB2) drains access liver copper via biological excretion in Wilson Disease rats>.
This study proves the efficacy of ARBM-101 in disease animal models based on copper emissions, an important requirement for determining whether Wilson's disease is treated or not.
In this paper, Arbormed demonstrated that administering ARBM-101 to mice that caused Wilson's disease induces rapid copper excretion compared to controls. It also confirmed the pharmacological effect of minimizing side effects by discharging copper through feces, a normal copper discharge route and physiological phenomenon.
Arbormed explained that the study proved that ARBM-101 is an innovative drug that can inhibit and treat the progression of hypertrophic cirrhosis caused by decreased liver function.
Arbormed previously released a study by the American Liver Society (AASLD) in 2022 that the administration of ARBM-101 to animal models has the effect of promoting the rapid discharge of copper while minimizing side effects. Professor Valentina Medici, UC Davis, one of the Arbormed advisors, was in charge of the presentation at the time.
In addition, it received attention from related academia by announcing the results of research on animal models for Wilson's disease at the "Wilson Aarthus 2022 Symposium," a major meeting related to Wilson's disease.
Arbormed plans to complete the ongoing ARBM-101 non-clinical study by Q1 2024. It will then enter the first phase of global clinical trials in the summer of 2024 and secure major data by the end of the year.
Wilson's disease is a congenital genetic disease that affects about 1 in 30,000 people, causing hepatitis symptoms, liver fibrosis, liver cirrhosis, and death in severe cases.
Penicillamine, approved by the FDA in 1970 as a treatment for Wilson's disease, is relatively less effective in relieving symptoms such as liver cirrhosis and has several side effects, with about 30% of patients stopping the drug. ARBM-101 is expected to improve major symptoms and is taken once a month.
Following the presentation of ARBM-101 animal model research data at major conferences last year, we recently established a clinical advisory committee for prominent medical staff in the United States. "As we have formed an advisory committee with world-class Wilson disease experts, we will speed up in-depth discussions on ARBM-101 clinical trials."
"We will submit a clinical trial plan (IND) to the US FDA (Food and Drug Administration) as soon as possible to enter the ARBM-101 clinical trial," said CEO Park Kyo-jin and "As ARBM-101 is a completely new treatment that has never existed before, we will do our best with the best scholars to develop it as an innovative new drug that can dramatically improve the quality of life of Wilson disease patients."
It is difficult to find a case in which this level of advisory committee has been formed to support clinical trials of new drug pipelines by domestic bio ventures. This is proof that overseas medical sites have high expectations for ARBM-101.
Arbormed recently published a study in an international journal demonstrating the safety and effectiveness of ARBM-101.
In March, the results of an animal model experiment of ARBM-101 were published in Gastroenterology (IF 33.883), a world-renowned journal in the field of digestive organs and the official journal of the American Society of Gastroenterology.
The title of this paper is <ARBM101 (Methanobactin SB2) drains access liver copper via biological excretion in Wilson Disease rats>.
This study proves the efficacy of ARBM-101 in disease animal models based on copper emissions, an important requirement for determining whether Wilson's disease is treated or not.
In this paper, Arbormed demonstrated that administering ARBM-101 to mice that caused Wilson's disease induces rapid copper excretion compared to controls. It also confirmed the pharmacological effect of minimizing side effects by discharging copper through feces, a normal copper discharge route and physiological phenomenon.
Arbormed explained that the study proved that ARBM-101 is an innovative drug that can inhibit and treat the progression of hypertrophic cirrhosis caused by decreased liver function.
Arbormed previously released a study by the American Liver Society (AASLD) in 2022 that the administration of ARBM-101 to animal models has the effect of promoting the rapid discharge of copper while minimizing side effects. Professor Valentina Medici, UC Davis, one of the Arbormed advisors, was in charge of the presentation at the time.
In addition, it received attention from related academia by announcing the results of research on animal models for Wilson's disease at the "Wilson Aarthus 2022 Symposium," a major meeting related to Wilson's disease.
Arbormed plans to complete the ongoing ARBM-101 non-clinical study by Q1 2024. It will then enter the first phase of global clinical trials in the summer of 2024 and secure major data by the end of the year.
Wilson's disease is a congenital genetic disease that affects about 1 in 30,000 people, causing hepatitis symptoms, liver fibrosis, liver cirrhosis, and death in severe cases.
Penicillamine, approved by the FDA in 1970 as a treatment for Wilson's disease, is relatively less effective in relieving symptoms such as liver cirrhosis and has several side effects, with about 30% of patients stopping the drug. ARBM-101 is expected to improve major symptoms and is taken once a month.
Following the presentation of ARBM-101 animal model research data at major conferences last year, we recently established a clinical advisory committee for prominent medical staff in the United States. "As we have formed an advisory committee with world-class Wilson disease experts, we will speed up in-depth discussions on ARBM-101 clinical trials."
"We will submit a clinical trial plan (IND) to the US FDA (Food and Drug Administration) as soon as possible to enter the ARBM-101 clinical trial," said CEO Park Kyo-jin and "As ARBM-101 is a completely new treatment that has never existed before, we will do our best with the best scholars to develop it as an innovative new drug that can dramatically improve the quality of life of Wilson disease patients."
관련링크
- 이전글Arbormed's "unique" Wilson disease mechanism, safety and effectiveness all at once 23.05.03
- 다음글BC World Pharm.co.,Ltd. and ArborMed Announce Research Collaboration Agreement on new drugs for "acute respiratory distress syndrome." 22.11.18
댓글목록
등록된 댓글이 없습니다.